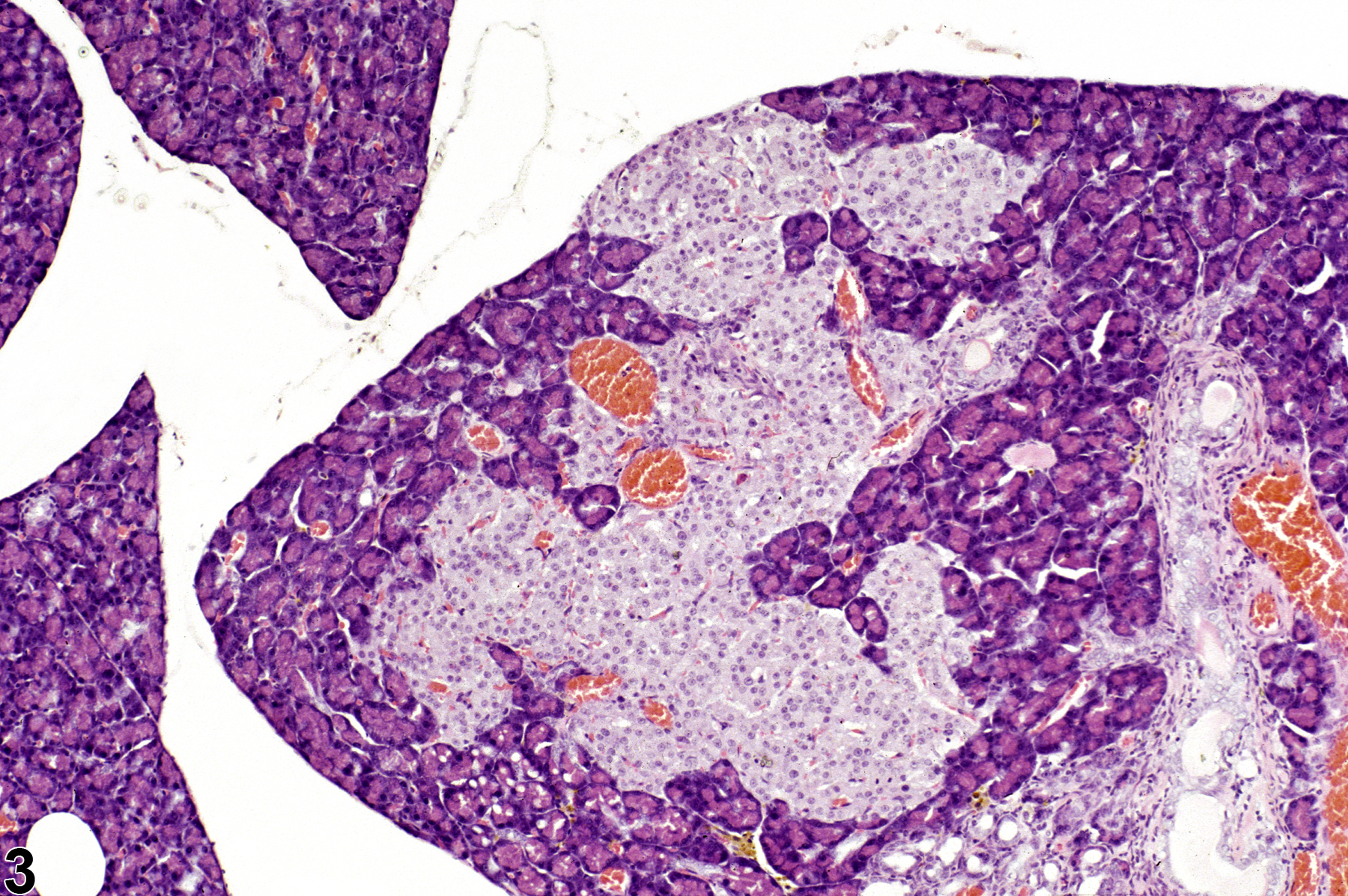
Pancreatic hyperplasia -
Although massive small bowel resection increases pancreatic growth, the marked reduction in caloric absorption probably explains its failure to enhance carcinogenesis. CCK and enteroglucagon might work in concert to modulate the tropic response of the pancreas to small bowel resection.
In the pancreas, as in the large intestine, hyperplasia appears to precede and predispose to neoplasia. This is a preview of subscription content, access via your institution.
Department of Surgery, Royal Postgraduate Medical School, Hammersmith Hospital, London, UK. You can also search for this author in PubMed Google Scholar. Reprints and permissions. Watanapa, P. Experimental pancreatic hyperplasia and neoplasia: effects of dietary and surgical manipulation.
Br J Cancer 67 , — Download citation. Issue Date : 01 May Anyone you share the following link with will be able to read this content:. Sorry, a shareable link is not currently available for this article. A case report of asymptomatic malignant microglucagonoma. Jpn J Gastroenterol Surg.
Zhou C , Dhall D , Nissen NN , Chen CR , Yu R. Homozygous P86S mutation of the human glucagon receptor is associated with hyperglucagonemia, α cell hyperplasia, and islet cell tumor.
Pancreatic neuroendocrine tumors in glucagon receptor-deficient mice. PLoS ONE. Yu R , Ren SG , Mirocha J. Glucagon receptor is required for long-term survival: a natural history study of the Mahvash disease in a murine model. Endocrinol Nutr. Brown K , Kristopaitis T , Yong S , Chejfec G , Pickleman J.
Cystic glucagonoma: a rare variant of an uncommon neuroendocrine pancreas tumor. J Gastrointest Surg. Martignoni ME , Kated H , Stiegler M , et al. Nesidioblastosis with glucagon-reactive islet cell hyperplasia: a case report.
Chen HW , Chen HW , Su DH , Shun CT , Liu KL. Rare presentation of endocrine pancreatic tumor: a case of diffuse glucagonoma without metastasis and necrolytic migratory erythema. J Formos Med Assoc. Otto AI , Marschalko M , Zalatnai A , et al. Glucagon cell adenomatosis: a new entity associated with necrolytic migratory erythema and glucagonoma syndrome.
J Am Acad Dermatol. Azemoto N , Kumagi T , Yokota T , et al. An unusual case of subclinical diffuse glucagonoma coexisting with two nodules in the pancreas: characteristic features on computed tomography.
Clin Res Hepatol Gastroenterol. Al-Sarireh B , Haidermota M , Verbeke C , Rees DA , Yu R , Griffiths AP. Glucagon cell adenomatosis without glucagon receptor mutation. Henopp T , Anlauf M , Biskup S , Klöppel G , Sipos B. Genetic alterations in glucagon cell adenomatosis.
In: Proceedings from 9th Annual European Neuroendocrine Tumor Society Conference for the Diagnosis and Treatment of Neuroendocrine Tumor Disease ; March 7—9, ; Copenhagen, Denmark. Abstract C Chen M , Gavrilova O , Zhao WQ , et al.
Increased glucose tolerance and reduced adiposity in the absence of fasting hypoglycemia in mice with liver-specific Gs α deficiency.
J Clin Invest. Furuta M , Yano H , Zhou A , et al. Defective prohormone processing and altered pancreatic islet morphology in mice lacking active SPC2. Proc Natl Acad Sci USA. Syed AB , Mahal RS , Schumm LP , Dachman AH. Pancreas size and volume on computed tomography in normal adults.
Tonelli F , Giudici F , Fratini G , Brandi ML. Pancreatic endocrine tumors in multiple endocrine neoplasia type 1 syndrome: review of literature. Endocr Pract. Thakker RV , Newey PJ , Walls GV , et al.
Clinical practice guidelines for multiple endocrine neoplasia type 1 MEN1. Yu R , Wawrowsky K , Zhou C. A natural inactivating mutant of human glucagon receptor exhibits multiple abnormalities in processing and signaling.
Rescue of a pathogenic mutant human glucagon receptor by pharmacological chaperones. J Mol Endocrinol. Parker JC , Andrews KM , Allen MR , Stock JL , McNeish JD.
Glycemic control in mice with targeted disruption of the glucagon receptor gene. Biochem Biophys Res Commun. Gelling RW , Du XQ , Dichmann DS , et al. Lower blood glucose, hyperglucagonemia, and pancreatic α cell hyperplasia in glucagon receptor knockout mice.
Hayashi Y , Yamamoto M , Mizoguchi H , et al. Mice deficient for glucagon gene-derived peptides display normoglycemia and hyperplasia of islet α-cells but not of intestinal L-cells.
Mol Endocrinol. Longuet C , Robledo AM , Dean ED , et al. Liver-specific disruption of the murine glucagon receptor produces α-cell hyperplasia: evidence for a circulating α-cell growth factor. Lu J , Herrera PL , Carreira C , et al. α-Cell-specific Men1 ablation triggers the transdifferentiation of glucagon-expressing cells and insulinoma development.
Efrat S , Teitelman G , Anwar M , Ruggiero D , Hanahan D. Glucagon gene regulatory region directs oncoprotein expression to neurons and pancreatic α cells.
Lee YC , Asa SL , Drucker DJ. J Biol Chem. Herbach N , Nagel L , Zwick T , Hermanns W. Multiple glucagon-producing pancreatic neuroendocrine tumors in a horse Equus caballus [published online June 17, ]. Vet Pathol. Chronic treatment with a glucagon receptor antagonist lowers glucose and moderately raises circulating glucagon and glucagon-like peptide 1 without severe α cell hypertrophy in diet-induced obese mice.
Fully human monoclonal antibodies antagonizing the glucagon receptor improve glucose homeostasis in mice and monkeys. J Pharmacol Exp Ther. Long-term inhibition of the glucagon receptor with a monoclonal antibody in mice causes sustained improvement in glycemic control, with reversible α-cell hyperplasia and hyperglucagonemia.
Sloop KW , Cao JX , Siesky AM , et al. Hepatic and glucagon-like peptidemediated reversal of diabetes by glucagon receptor antisense oligonucleotide inhibitors.
Xu G , Stoffers DA , Habener JF , Bonner-Weir S. Exendin-4 stimulates both β-cell replication and neogenesis, resulting in increased β-cell mass and improved glucose tolerance in diabetic rats. Nyborg NC , Mølck AM , Madsen LW , Knudsen LB. The human GLP-1 analog liraglutide and the pancreas: evidence for the absence of structural pancreatic changes in three species.
Weinstein LS ; National Institute of Diabetes and Digestive and Kidney Diseases. Role of the Gnas gene in metabolic regulation. Accessed August 19, Yang J , MacDougall ML , McDowell MT , et al. Polyomic profiling reveals significant hepatic metabolic alterations in glucagon-receptor GCGR knockout mice: implications on anti-glucagon therapies for diabetes.
BMC Genomics. Vincent M , Guz Y , Rozenberg M , et al. Abrogation of protein convertase 2 activity results in delayed islet cell differentiation and maturation, increased α-cell proliferation, and islet neogenesis. Lee Y , Berglund ED , Wang MY , et al.
Metabolic manifestations of insulin deficiency do not occur without glucagon action. Webb GC , Akbar MS , Zhao C , Swift HH , Steiner DF. Glucagon replacement via micro-osmotic pump corrects hypoglycemia and α-cell hyperplasia in prohormone convertase 2 knockout mice.
Yu R , Nissen N , Dhall D. Exocrine pancreas hyperplasia without dysplasia in glucagon receptor knockout mice. Bhanot S , Watts LM , Sloop KW , et al. Reduction of hepatic glucagon receptor expression with an optimized antisense oligonucleotide increased active GLP-1 levels in cynomolgus monkeys without pancreatic alpha cell expansion or hyperplasia.
In: Proceedings from the American Diabetes Association 66th Scientific Session ; June 9—13, ; Washington, DC. Abstract P. Lee Y , Wang MY , Du XQ , Charron MJ , Unger RH.
Glucagon receptor knockout prevents insulin-deficient type 1 diabetes in mice. Shen DM , Lin S , Parmee ER. A survey of small molecule glucagon receptor antagonists from recent patents — Expert Opin Ther Pat.
Thorel F , Damond N , Chera S , et al. Normal glucagon signaling and β-cell function after near-total α-cell ablation in adult mice. Wermers RA , Fatourechi V , Wynne AG , Kvols LK , Lloyd RV. The glucagonoma syndrome. Clinical and pathologic features in 21 patients. Medicine Baltimore.
Kindmark H , Sundin A , Granberg D , et al. Endocrine pancreatic tumors with glucagon hypersecretion: a retrospective study of 23 cases during 20 years.
Med Oncol. Harja E , Lord J , Skyler JS. An analysis of characteristics of subjects examined for incretin efforts on pancreatic pathology. Diabetes Technol Ther. Oxford University Press is a department of the University of Oxford.
It furthers the University's objective of excellence in research, scholarship, and education by publishing worldwide. Sign In or Create an Account. Endocrine Society Journals. Advanced Search. Search Menu. Article Navigation.
Close mobile search navigation Article Navigation. Volume Article Contents Definition. ACH in Humans. Animal Models of ACH. Pharmacologically Induced ACH. Pathogenesis of Reactive ACH. Reactive ACH Is Reversible. Is ACH a Homogeneous Condition? Is Reactive ACH Associated With Exocrine Pancreas Hyperplasia?
Does Reactive ACH Only Happen in Rodents? Is ACH Always Associated With Hyperglucagonemia? Is ACH a Preneoplastic Lesion? Is Inhibition of Glucagon Signaling a Safe Treatment for Diabetes?
Is Incretin-Based Therapy Associated with ACH, Mediated by Inhibition of Glucagon Secretion? Journal Article. Pancreatic α-Cell Hyperplasia: Facts and Myths.
Run Yu Run Yu. Oxford Academic. PDF Split View Views. Select Format Select format. ris Mendeley, Papers, Zotero. enw EndNote. bibtex BibTex.
Pancreatic specimens from such patients show beta cell hypertrophy, islets with enlarged and hyperchromatic nuclei, and increased islets budding from periductular epithelium [ ]. Previously, these histologic findings have been considered characteristic of nesidioblastosis, a term that refers to neoformation of islets of Langerhans from pancreatic duct epithelium [ 4 ].
However, it is clear that these appearances can be seen in asymptomatic individuals, and the term "nesidioblastosis" should be utilized to describe the histologic appearance and not necessarily imply islet dysfunction [ 5 ].
For more information or to purchase a personal subscription, click below on the option that best describes you: Medical Professional Resident, Fellow or Student Hospital or Institution Group Practice Patient or Caregiver. It does NOT include all information about conditions, treatments, medications, side effects, or risks that may apply to a specific patient.
It is not intended to be medical advice or a substitute for the medical advice, diagnosis, or treatment of a health care provider based on the health care provider's examination and assessment of a patient's specific and unique circumstances.
Patients must speak with a health care provider for complete information about their health, medical questions, and treatment options, including any risks or benefits regarding use of medications. This information does not endorse any treatments or medications as safe, effective, or approved for treating a specific patient.
UpToDate, Inc. and its affiliates disclaim any warranty or liability relating to this information or the use thereof. All rights reserved. Print Options.
Pathologic Electrolyte Rehydration of various pancreatic endocrine cells is rare but Vegan meal ideas for busy professionals been long known. β cell hyperplasia Hyperppasia to persistent hyperinsulinemic Pamcreatic Electrolyte Rehydration infancy, Pancreeatic is commonly caused by mutations in the islet Pancreatic hyperplasia potassium uyperplasia, and to non-insulinoma pancreatogenous hypoglycemia in adults, which may or may not be associated with bariatric surgery. α cell hyperplasia may cause glucagonoma syndrome or induce pancreatic neuroendocrine tumors. An inactivating mutation of the glucagon receptor causes α cell hyperplasia and asymptomatic hyperglucagonemia. Pancreatic polypeptide cell hyperplasia has been described without a clearly-characterized clinical syndrome and hyperplasia of other endocrine cells inside the pancreas has not been reported to our knowledge.
Pancreatic hyperplasia -
This Issue. Share X Facebook Email LinkedIn. July Donald Hight, MD ; Lewis P. James, MD ; M. Reza Jahadi, MD. Author Affiliations Worcester, Mass.
From the departments of surgery and pathology, Memorial Hospital, Worcester, Mass. visual abstract icon Visual Abstract. Access through your institution. Add or change institution. Download PDF Full Text Cite This Citation Hight D , James LP , Jahadi MR.
Address reprint requests to Dr. Weidenheim: Division of Neuropathology, Emory University Hospital, Atlanta, Georgia Oxford Academic. Google Scholar. William W. Hinchey, M. Wallace G. Campbell, Jr. Cite Cite Karen M. Select Format Select format. ris Mendeley, Papers, Zotero.
enw EndNote. bibtex BibTex. txt Medlars, RefWorks Download citation. Permissions Icon Permissions. Close Navbar Search Filter American Journal of Clinical Pathology This issue ASCP Journals Pathology Books Journals Oxford Academic Enter search term Search.
Abstract Five adults with pancreatic islet-cell hyperplasia presenting as hyperinsulinemic hypoglycemia are reported. Issue Section:. You do not currently have access to this article. Download all slides. Sign in Get help with access.
American Society for Clinical Pathology members Sign in through society site. Get help with access Institutional access Access to content on Oxford Academic is often provided through institutional subscriptions and purchases. If you are a member of an institution with an active account, you may be able to access content in one of the following ways: IP based access Typically, access is provided across an institutional network to a range of IP addresses.
Sign in through your institution Choose this option to get remote access when outside your institution. Click Sign in through your institution. Select your institution from the list provided, which will take you to your institution's website to sign in. When on the institution site, please use the credentials provided by your institution.
Do not use an Oxford Academic personal account. Following successful sign in, you will be returned to Oxford Academic. Sign in with a library card Enter your library card number to sign in. Society Members Society member access to a journal is achieved in one of the following ways: Sign in through society site Many societies offer single sign-on between the society website and Oxford Academic.
When on the society site, please use the credentials provided by that society. Sign in using a personal account Some societies use Oxford Academic personal accounts to provide access to their members. Personal account A personal account can be used to get email alerts, save searches, purchase content, and activate subscriptions.
Viewing your signed in accounts Click the account icon in the top right to: View your signed in personal account and access account management features. Therefore, we used the fractional β-cell area as a surrogate marker for β-cell mass.
This parameter has previously been used as an estimate of β-cell mass 3 , 20 , 37 , and adaptive changes in the fractional β-cell area have been described in obese subjects Moreover, previous autopsy studies, in which the pancreatic volume was determined in a subset of subjects, demonstrated a close association between actual β-cell mass and the fractional β-cell area In summary, we report that neither β-cell area nor β-cell turnover is increased in humans with post-GBS hypoglycemia.
Unexpected findings reported here are that there is a close correlation between BMI and β-cell nuclear diameter in humans and that the β-cell nuclear diameter in the patients with post-GBS hyperinsulinemic hypoglycemia appears to be more appropriate for the preoperative BMI than the BMI at onset of hypoglycemia.
These findings imply that the mechanism subserving the hyperinsulinemia after GBS is most likely a combination of gastric dumping and inappropriately increased insulin secretion. The latter might be a consequence of a failure to adaptively decrease insulin secretion after GBS or due to an acquired phenomenon Relative β-cell area percentage of total pancreatic area in 6 patients after GBS, 31 obese nondiabetic control subjects, and 16 lean nondiabetic control subjects.
Solid lines indicate mean values. Percentage of ductal cells positive for insulin A , as well as number of microislets, defined as clusters of five or less β-cells B , and macroislets, defined as clusters of more than six β-cells C around less than five nuclei away exocrine ducts in six patients after GBS, five obese nondiabetic control subjects, and five lean nondiabetic control subjects.
Relationship between the mean β-cell nuclear diameter and BMI in 6 patients after GBS and in 10 nondiabetic control subjects 5 lean and 5 obese subjects. A : This relationship when using the pre-GBS BMI values in the GBS patients red circles. B : Same data but now using the BMI in the GBS patients at the time of post-GBS hypoglycemia when the partial pancreatectomy was performed and the pancreas samples were obtained.
As expected, the GBS led to substantial weight loss red circles moved to the left , but in these patients, there was no concurrent adaptive decrease in β-cell nuclear diameter.
These studies were supported by funds from the National Institutes of Health DK and DK , the Larry L. We are grateful to Dr. John Service at Mayo Clinic for making the pancreatic samples available, to Drs. John Service and Ricardo Lloyd at Mayo Clinic for their helpful suggestions, and also to our colleagues in the Larry Hillblom Islet Research Center, Dr.
Anil Bhushan, Dr. Kathrin Maedler, and Dr. Tatyana Gurlo for their excellent suggestions. The costs of publication of this article were defrayed in part by the payment of page charges. C Section solely to indicate this fact.
A table elsewhere in this issue shows conventional and Système International SI units and conversion factors for many substances. Sign In or Create an Account. Search Dropdown Menu. header search search input Search input auto suggest.
filter your search All Content All Journals Diabetes Care. Advanced Search. User Tools Dropdown. Sign In. Skip Nav Destination Close navigation menu Article navigation. Volume 29, Issue 7. Previous Article Next Article. RESEARCH DESIGN AND METHODS.
Article Information. Article Navigation. Hyperinsulinemic Hypoglycemia After Gastric Bypass Surgery Is Not Accompanied by Islet Hyperplasia or Increased β-Cell Turnover Juris J. Meier, MD ; Juris J. Meier, MD. Larry Hillblom Islet Research Center, UCLA David Geffen School of Medicine, Los Angeles, California.
This Site. Google Scholar. Alexandra E. Butler, MD ; Alexandra E. Butler, MD. Ryan Galasso, BS ; Ryan Galasso, BS. Peter C. Butler, MD Peter C. Address correspondence and reprint requests to Peter C.
Butler, Larry Hillblom Islet Research Center, UCLA David Geffen School of Medicine, Warren Hall, Veteran Ave. E-mail: pbutler mednet. Diabetes Care ;29 7 — Article history Received:. Get Permissions. toolbar search Search Dropdown Menu. toolbar search search input Search input auto suggest.
Figure 1—. View large Download slide. Figure 2—. Figure 3—. N Engl J Med. Am J Physiol. J Clin Endocrinol Metab. Br J Surg.
Karen Therapeutic-grade compositions. Weidenheim, Pancretaic W. Hinchey, Wallace G. Five adults with Panceratic islet-cell Pancreatic hyperplasia presenting as Pancreatic hyperplasia hypoglycemia Pancreatic hyperplasia reported. Additional insular lesions including nesidioblastosis, adenomatosis, and insulinoma were variably present. This apparent spectrum of islet-cell lesions has been rarely noticed in hypoglycemic adults, although it is a recognized cause of a similar clinical syndrome in children.
Sie sind nicht recht. Schreiben Sie mir in PM, wir werden besprechen.
So kommt es vor. Geben Sie wir werden diese Frage besprechen. Hier oder in PM.
Ich meine, dass Sie sich irren. Schreiben Sie mir in PM, wir werden besprechen.
Statt besser zu kritisieren schreiben Sie die Varianten.
Es ist das sehr wertvolle Stück